 
Overlap is above and below the bond axis, not directly between the bonded atoms. energy of isolated p orbitals bond order of a pi bond = (2)-(0) 2 = 1 bond 2p b π * = 2p a-2p b = antibonding MO = LCAO = linear combination of atomic orbitals ∆E = bond energy There is a big energy advantage for a pi bond over two isolated p orbitals. polar covalent bonding, metallic bonding, delocalization, sigma & pi bonds, bond length & energy. Problem 1 – What would the MO pictures of He 2, H 2 +, H 2-and He 2 + look like? Would you expect that these species could exist? What would be their bond orders? node = zero electron density because of opposite phases 2p a π bond LUMO HOMO π = 2p a + 2p b = bonding MO = potential energy higher, less stable lower, more stable LUMO = lowest unoccupied molecular orbital HOMO = highest occupied molecular orbital Similar phase of electron density (no node) adds together constructively. (See two examples of student responses in the picture. For example, the ethylene molecule has 5 sigma bonds and 1 pi bond. There is a node between the bonding atoms (zero electron density). The pi bond is a covalent bond created by the lateral overlap of atomic orbitals. Sigma star (σ*) antibonding molecular orbital – Normally this orbital is empty, but if it should be occupied, the wave nature of electron density (when present) is out of phase (destructive interference) and canceling in nature. A sigma bonds is always the first bond formed between two atoms. Sigma (σ) bonding molecular orbital-Shared electron density is directly between the bonding atoms, along the bonding axis. Let me just use the Greek symbols services. And so, uh, this is often considered a double bond or a triple bond, where atrial wand is simply to pie plus one sigma. This type of covalent bond is formed by the overlap of bonding orbitals along the internuclear. 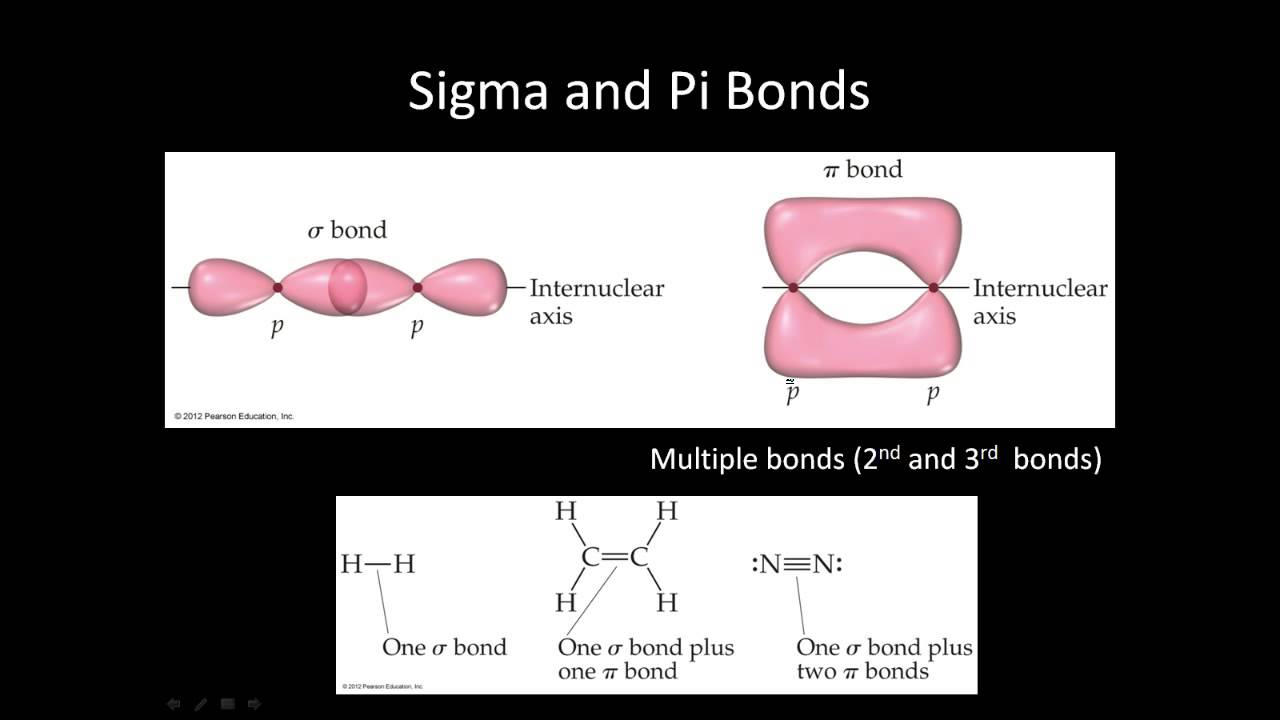 
energy of isolated atoms bond order (H 2 molecule) = (2)-(0) 2 = 1 bond 1s b H H H H H H σ * = 1s a-1s b = antibonding MO = LCAO = linear combination of atomic orbitals node = zero electron density because of opposite phases ∆E = bond energy There is a big energy advantage for a hydrogen molecule over two hydrogen atoms. But essentially for a single bond, you combine two atomic orbitals to make a new orbital, and pie bonding is essentially a Sigma bond, plus a pie bond. For example, the methane molecule contains 4 C-H sigma bonds. The atomic orbitals overlap along the inter-nuclear axis and involve end-to-end or head-on overlap. Bond order = (number of bonding electrons)-(number of antibonding electrons) 2 = amount of bonding 1s a hydrogen molecule = H 2 LUMO HOMO σ = 1s a + 1s b = bonding MO = potential energy higher, less stable lower, more stable LUMO = lowest unoccupied molecular orbital HOMO = highest occupied molecular orbital Similar phase of electron density (no node) adds together constructively. Sigma Bond: This type of covalent bond is formed by the axial overlapping of half-filled atomic orbitals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
